Science News for Vascular Discovery 2020
Important Links
Vascular Discovery 2020 From Genes to Medicine Welcome
Mary Sorci-Thomas, PhD, FAHA chair of the council on Arteriosclerosis, Thrombosis, and Vascular Biology, and Lars Maegdefessel, MD, PhD welcome viewers to Vascular Discovery 2020 and discuss the importance of continuing to share the latest in vascular research even in the midst of a world-wide pandemic.
Thrombosis and Cancer | Nigel Mackman, PhD, FAHA
Vascular Discovery 2020 Program Committee Co-Chair Katey Rayner, PhD, FAHA interviews Nigel Mackman, PhD, FAHA about several lines of research exploring the connection between thrombosis and cancer. Mackman presented his research at Tuesday afternoon's plenary session.
Single Cell RNA Sequencing Reveals Heterogeneous Smooth Muscle Cell Phenotype Modulation in Marfan Syndrome Aortic Aneurysm
Albert J Pedroza | Stanford University, Palo Alto, CA
Results: Single cell RNA sequencing finds smooth muscle cell changes in Marfan syndrome aortic aneurysm.
Quantitative Trait Loci Mapped For Tcf21 Binding, Chromatin Accessibility And Chromosomal Looping In Coronary Artery Smooth Muscle Cells Reveal Molecular Mechanisms Of Coronary Disease Loci

Quanyi Zhao | Stanford University, Stanford, CA
Results: Signaling in vascular smooth muscle cells has implications for vascular malformations.
Endothelial Deficiency of Nogo-B Receptor Induces Cerebral Hemorrhage via Histone Acetylation Regulated Ccm1/2 Expression

Qing Robert Miao | NYU Winthrop Hospital, Mineola, NY
Results: Possible mechanism described for how endothelial deficiency of nogo-B receptor may contribute to sporadic cerebral cavernous malformation disease.
Awards presented Tuesday
Daniel Steinberg Early Career Investigator Award in Atherosclerosis/Lipoproteins

Hiroaki Okazaki | Tokyo, Japan
Karl Link Early Career Investigator Award in Thrombosis

Clot contraction drives the translocation of procoagulant platelets to thrombus surface
Dmitry Y. Nechipurenko, PhD | Lomonosov Moscow State University, Moscow, RU
Werner Risau Early Career Investigator Award in Vascular Biology

Ponatinib Combined with Rapamycin Causes Regression of Murine Venous Malformation
Elisa Boscolo, PhD, Cincinnati Children's Hospital Medical Center
2020 Mentor of Women Award

Robert W Hobson MD Early Career Investigator
Circulating Fatty Acid Synthase And Diabetes Are Independently Associated With Chronic Limb Threatening Ischemia
Shirli Tay | Washington University School of Medicine
Junior Investigator Award for Women
Loss of Notch Signaling in Pericytes Induces Arteriovenous Malformations
Taliha Nadeem | University of Illinois at Chicago, Chicago, IL
Thrombosis and Tissue Specific Hemostasis | Maureane Hoffman, MD, PhD
Maureane Hoffman, MD, PhD describes her recent work on thrombosis and tissue-specific hemostasis which she presented Thursday during Vascular Discovery 2020.
Epigenetic Regulation of the Notch Signaling Pathway Modulates Macrophage/T Cell Phenotype and Impairs Diabetic Wound Healing

Frank Michael Davis | University of Michigan, Ann Arbor, MI
Results: Epigenetic regulation of the notch signaling pathway modulates macrophage/T cell phenotype and impairs diabetic wound healing.
Immune Checkpoint Inhibitors Aggravate T Cell-mediated Plaque Inflammation in Experimental Atherosclerosis

Kikkie Poels | Amsterdam UMC, Amsterdam, Netherlands
Results: Immune checkpoint inhibitors aggravate T cell-mediated plaque inflammation in experimental atherosclerosis.
Brain Emotional Activity is Associated With Acute Plaque Instability via Enhanced Macrophage Activity: A Prospective 3d Rendered 18f Fdg-pet/ct Assessment

Dong Oh Kang | Multimodal Imaging and Theranostic Lab, Cardiovascular Ctr, Korea Univ Guro Hosp, Seoul, Korea, Republic of
Results: Findings suggest brain emotional activity is associated with acute plaque instability via enhanced macrophage activity.
News release: Brain emotional activity linked to blood vessel inflammation in recent heart attack patients
The Phosphatidylserine Receptor Tim4 and Integrin Beta 5 Subunit are Required for Effective Lysosomal Secretion in Macrophage Clearance of Apoptotic Adipocytes
Valeria Barbosa-Lorenzi | Weill Cornell Med, New York, NY
Results: Identification of major pathways for lysosomal secretion in macrophage clearance of apoptotic adipocytes may lead to treatments for T2DM, cancers, or metabolic disorders.
Interrogation of Long Intergenic Non-coding RNA Functions in Obesity-induced Adipose Tissue Inflammation
Marcella E O'Reilly | Columbia University Medical Ctr, New York, NY
Results: A novel lincRNA may regulate obesity-induced adipose tissue inflammation; understanding more may lead to new therapies.
Targeting Myeloid-cell Specific Integrin Alpha9Beta1 Inhibits Arterial Thrombosis and Improves Stroke Outcome by Limiting Thromboinflammation
Nirav Dhanesha | University of Iowa, Iowa City, IA
Results: Target and mechanism identified to inhibit arterial thrombosis and improve stroke outcomes.
Vein-on-chip: Microengineered Modeling of Venous Valves, Thrombosis and Therapeutics
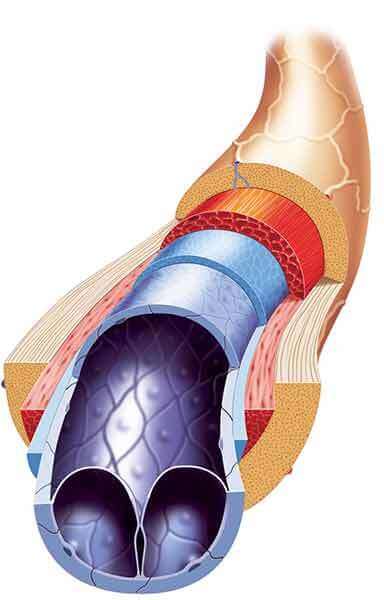
Abhishek Jain, Texas A&M University, College Station, TX
Results: Vein-Chip models venous valves, thrombosis processes, and treatments.
News release: Miniature version of human vein allows study of deep vein thrombosis
Winner: Evaluating the Impact of Thrombocytopenia and Platelet Transfusion on Bleeding and Thrombosis in the Setting of Anti-Platelet Therapy
Robert H Lee | University of North Carolina, Chapel Hill, NC
Results: Study informs about DAPT safety in thrombocytopenia and best use of platelet transfusion for bleeding reversal.
A Genome-Wide Screen for Modifiers of Tissue Factor Activity Identifies the Palmitoyltransferase ZDHHC2 as a Negative Regulator of Coagulation Initiation
Sol Schulman | Beth Israel Deaconess Medical Ctr, Boston, MA
Results: Genome-wide screen identifies a negative regulator of coagulation initiation.
Irvine H. Page Junior Faculty Research Award Finalists
Adipocyte Tribbles1 Regulates Plasma Lipids and Circulating Adiponectin

Robert C Bauer | Columbia University, New York, NY
Results: Novel role for TRIB1 identified in plasma lipid metabolism regulation.
H3K4 Di-methylation Controls Vascular Smooth Muscle Cell Lineage Identity, Contractility, and Participation in Vascular Remodeling
Delphine A Gomez | University of Pittsburgh, Pittsburgh, PA
Results: Mechanism studied for regulation of smooth muscle cell participation in vascular remodeling.
Endothelial Rna-binding Protein Elavl1 (HuR) Regulates Alternative Splicing of Genes Involved in Adaptive Immunity and Increases T Cells in the Atherosclerotic Intima

Patrick A Murphy | UConn Health, Farmington, CT
Results: Endothelial RNA-binding protein may play a key role in atherosclerosis.
Winner: A Genome-Wide CRISPR Screen in Primary Macrophages Reveals WDFY3 as a Novel Regulator of Macrophage Efferocytosis in vitro and in vivo

Hanrui Zhang | Columbia University Irving Medical Ctr, New York, NY
Results: A genome-wide CRISPR screen in primary macrophages reveals WDFY3 as a novel regulator of macrophage efferocytosis in vitro and in vivo.
Alan T Hirsch Mid-Career Award in Vascular Medicine
Design And Delivery Of Microrna Switches To Treat Cardiovascular Diseases
Hana Totary-jain | University of South Florida, Tampa, FL
Exploring Resilience to Cardiovascular Insults | Luisa Iruela Arispe, PhD
Vascular Discovery 2020 Invited Lecturer Louisa Iruela-Arispe, PhD describes some of her recent work exploring the molecular mechanisms contributing to recovery after cardiovascular injury.
Invited Lecture Series
Jeffrey M. Hoeg ATVB Basic Science and Clinical Research Award

Elena Aikawa, MD, PhD, FAHA is Professor of Medicine at Harvard Medical School. She is Co-director of the Center for Interdisciplinary Cardiovascular Sciences, Founding Director of the Heart Valve Translational Research Program and Associate Head of Cardiovascular Life Sciences Section at Brigham and Women’s Hospital.
Her research focuses on the development of new therapies to prevent and treat calcific aortic valve stenosis, a disease that currently has no treatment options except invasive valve replacement. She was at the forefront in the discovery of inflammation-dependent mechanisms of cardiovascular calcification. Her studies contributed to the discovery of calcifying extracellular vesicles - precursors of microcalcification (Circulation Research 2013; Nature Materials 2016, JCI 2016). Recently she used systems approaches, involving multi-omics and network medicine, to identify novel anti-calcification targets (Circulation 2018).
Dr. Aikawa was a member of the NHLBI Working Group on Calcific Aortic Valve Stenosis and the Alliance of Investigators on Calcific Aortic Valve Disease. She holds editorial board member positions at PLOS ONE, Journal of Extracellular Vesicles, Circulation Research and ATVB. Dr. Aikawa has delivered over 160 invited talks and authored more than 210 manuscripts. Her research program has been continuously supported by NIH funding.
In addition to her strong interest in research, Dr. Aikawa enjoys mentoring young scientists and is committed to advancing the careers of women in science and medicine. She is vice-chair of the ATVB Council Women's Leadership Committee and president of the International Society for Applied Cardiovascular Biology. She is serving as the society's first female president.
Distinguished Lecture

Joseph Loscalzo, MD, PhD, FAHA is Hersey Professor of the Theory and Practice of Medicine, and Soma Weiss, M.D., Distinguished Chair in Medicine at Harvard Medical School, Chairman of the Department of Medicine, and Physician-in-Chief at Brigham and Women’s Hospital. Dr. Loscalzo received his A.B. degree, summa cum laude, his Ph.D. in biochemistry, and his M.D. from the University of Pennsylvania. His clinical training was completed at Brigham and Women’s Hospital and Harvard Medical School, where he served as Resident and Chief Resident in medicine and Fellow in cardiovascular medicine. After completing his training, Dr. Loscalzo joined the Harvard faculty and staff at Brigham and Women’s Hospital in 1984. He rose to the rank of Associate Professor of Medicine, Chief of Cardiology at the West Roxbury Veterans Administration Medical Center, and Director of the Center for Research in Thrombolysis at Brigham and Women’s Hospital. He joined the faculty of Boston University in 1994, first as Chief of Cardiology and, in 1997, Wade Professor and Chair of Medicine, Professor of Biochemistry, and Director of the Whitaker Cardiovascular Institute. He returned to Harvard and Brigham and Women’s Hospital in 2005.
Dr. Loscalzo is recognized as an outstanding cardiovascular scientist, clinician, and teacher. He has received many awards, including the Clinician-Scientist Award, the Distinguished Scientist Award, the Research Achievement Award, the Paul Dudley White Award and the Gold Heart Award from the American Heart Association. He is currently Director of the NIH-funded Center for Accelerated Innovation (the Boston Biomedical Innovation Center), and of the NIH-funded Harvard Undiagnosed Disease Network program. He is also former Editor-in-Chief of Circulation, currently Editor-at-Large of the New England Journal of Medicine, and a current senior editor of Harrison’s Principles of Internal Medicine.
Dr. Loscalzo has been a visiting professor at many institutions, holds three honorary degrees, has authored or co-authored over 1,000 scientific publications, has authored or edited 51 books, and holds 32 patents for his work in the field of nitric oxide, redox biology, and vascular biology. He is also the recipient of many grants from the NIH and industry for his work in the areas of vascular biology, thrombosis, atherosclerosis, and, more recently, systems biology over the past thirty years. His most recent work has established the field of network medicine, a paradigm-changing discipline that seeks to re-define disease and therapeutics from an integrated perspective using systems biology and network science.